Editor’s Note – This essay was subsequently published as:
Dillon, R.T., Jr. (2023b) CPP Diary: The Spurious Lithasia of Caney Fork. Pp 9 - 15 in The Freshwater Gastropods of North America Volume 6, Yankees at The Gap, and Other
Essays. FWGNA Project, Charleston, SC.
The Duck River is our Galapagos, and Calvin Goodrich our
Darwin. Or possibly our
inverse-Galapagos, and our anti-Darwin, I’m not sure. For the brilliant evolutionary insight that
Goodrich glimpsed in 1934 through the lens of this rich, fresh waterway coursing
through the heart of middle Tennessee was not more species, but less.
It’s an old, old story [1], but let’s tell it again. Prior to the dawn of the modern synthesis,
North American freshwater malacology recognized at least 12 – 15 species of
pleurocerids in the Duck River, probably more.
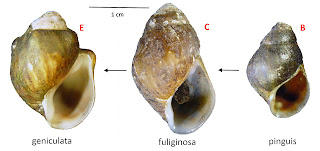
The lower reaches of the Duck (e.g., Wright Bend site E) were inhabited
by heavily-shelled, “obese” populations identified as Lithasia geniculata,
distinguished by their shells with prominently-shouldered whorls. The slightly-less-obese, smooth-shouldered
populations of the middle reaches (e.g., US41A, at site C) were identified as
Lithasia fuliginosa. And the headwaters
of the Duck River (e.g., Old Fort site B) were inhabited by Anculosa pinguis,
lightly-shelled populations without any shoulders on their whorls at all.
 |
| CPP in the Lithasia geniculata of the Duck River |
In 1934, Goodrich published his “Studies of the Gastropod
Family Pleuroceridae I,” in which he synonymized all these forms as subspecies
under L. geniculata [2]. He just did it,
at the top of the section, without making any sort of declaration, or using any
form of the noun “synonym,” as though his unique insight were already an
article of established malacological doctrine [3]. He then meticulously documented, town to town
and bridge to bridge down the length of the Duck, the gradual transition of the
three subspecies from one form to the next.
Even to me, his latter-day apostle, Goodrich’s 1934
intuition about the plasticity of shell phenotype in freshwater gastropods was
startlingly profound. His “Studies in
the Gastropod Family Pleuroceridae” series inspired me to coin the term
“Goodrichian taxon shift” in his honor in 2007, subsequently generalized to
cryptic phenotypic plasticity (CPP) by Dillon, Jacquemin and Pyron in 2013 [4].
So, six years later, Goodrich published a paper reviewing
the taxonomy and distribution of the pleurocerid fauna of the Ohio River
drainage in its entirety, not just the Lithasia of the Duck River but all
species in all seven genera in a vast region touching 14 states [5]. And the only other population of Lithasia
geniculata pinguis of which he was aware inhabited the Caney/Collins drainage
of the Cumberland River, the headwaters of which interdigitate with the Duck
immediately to the east, in the vicinity of McMinnville.
But alas, even in Goodrich’s day the diverse pleurocerid fauna of the Tennessee/Cumberland was rapidly disappearing in the face of impoundment, canalization, and widespread development for navigation and hydropower across the southern interior. The Caney/Collins system was terribly impacted by the impoundment of Center Hill Lake in the late 1940s, and the same almost happened in the Duck River in the 1970s, themes to which we shall return. Motivated by longstanding conservation concerns, in 2003 our colleagues Russ Minton and Chuck Lydeard undertook to construct a gene tree for the North American genus Lithasia [7].
Russ and Chuck a good job rounding up
samples from 11 of the species and subspecies of Lithasia listed by
Goodrich/Burch, 25 populations in all, sequencing in some cases as many as 6
individuals per population. From the
Duck River Russ and Chuck sequenced one population of L geniculata geniculata
(1 individual), three populations of L. geniculata fuliginosa (1, 3, and 3
individuals), and one population of L. geniculata pinguis (6 individuals). And they also included 2 individuals of
nominal Lithasia geniculata pinguis from a Caney/Collins population. And here is their gene tree:
 |
| Minton & Lydeard [7] Figure 3, modified. |
By now my readership will understand gene trees are
dependent variables, not independent variables [8]. You cannot work out the evolution of a set of
organisms from a gene tree. But if you
have developed an evolutionary hypothesis from good solid data of some broader
sort, you may be able to understand what a gene tree is telling you.
To completely unpack the message being telegraphed to us by
the enigmatic arboreal specimen figured above would require at least 6 – 8 blog
posts of standard length [9]. But for
the present let us focus on just the two little branches labelled “geniculata
pinguis” that I have circled in red. The
two sequences obtained from the 6 individuals sampled from the Duck River, D1
and D2, cluster with all the other Lithasia.
And the two sequences obtained from the Caney/Collins system, C1 and C2,
are way off with pleurocerids of other genera.
To quote Minton & Lydeard verbatim: “Further work needs to be
undertaken to determine the identity and placement of the Collins River taxa.”
Thanks, Captain Obvious! If Calvin Goodrich had enjoyed access to collections from
the Caney/Collins system of the same quality and detail that he enjoyed for the
Duck, he might well have recognized a gradual progression in the shell
phenotype of Pleurocera simplex quite analogous to that he documented for
Lithasia geniculata in 1934.
Most of the headwaters of Caney Fork and its tributaries
(e.g., site J) are inhabited by rather typical-looking populations of the
widespread Pleurocera simplex simplex, no different from those one might find
in tributaries of the Holston River around Saltville, VA, from whence the
species was described by Thomas Say in 1825 [11]. We featured the P. simplex population
inhabiting Pistol Creek at Maryville, TN in a series of essays published in 2016 [12], and the P.
simplex population inhabiting Gap Creek, TN, last month [13]. All of these populations are darkly
pigmented, and bear gracile, teardrop-shaped shells such as shown in figure J
below.
And all those populations inhabit small creeks and streams
primarily of groundwater. In East
Tennessee, populations of Pleurocera simplex do not typically extend into
larger rivers [14].
But in tributaries of the Cumberland, Kentucky, and Green
Rivers, Pleurocera simplex populations often do extend into rivers of
substantial size – as long as the currents are good and the rocky substrate they require does not
entirely give way to mud. Here their
shells become heavier, chunkier, and more lightly-pigmented. Goodrich [5] identified paler,
heavier-shelled populations such as are found in the Collins River at site K as
“Goniobasis ebenum (Lea 1841).”
And in the largest rivers of the Caney/Collins system (e.g.,
site L), populations of P. simplex are so robustly shelled that they can easily
be confused for Lithasia geniculata pinguis.
I speculate it may have been a sample of P. simplex that Minton & Lydeard
collected from the Collins River back in 2003, demonstrating intrapopulation
morphological variance so extreme as to prompt an (erroneous) identification of
Lithasia.
To be clear. Bona
fide populations of Lithasia geniculata pinguis do indeed inhabit the
Caney/Collins system, as Goodrich stated.
But they are typically sparse, and often swamped by dense populations of
P. simplex. Pleurocera simplex
populations do not range west into the Duck River drainage, however, and so
confusions of this sort did not complicate the story Goodrich told us in 1934.
So broadening the subject out through the rest of Middle Tennessee and into Kentucky. What is this enigmatic taxon described by Isaac Lea in 1841, "Melania" (aka Goniobasis, aka
Elimia, aka Pleurocera) ebenum? Goodrich [5] identified
ebenum populations through most of the Cumberland River drainage, from
“Cumberland River above the falls” through “Smith’s Shoals, Pulaski County,
Kentucky” west beyond Nashville to “springs and small streams” in Dickson
County, Tennessee. Could all these populations that Goodrich called "Goniobasis ebenum" be pale, triangular, robustly-shelled P.
simplex? Stay tuned.
Notes
[1] The best entry into this literature would be to purchase
FWGNA Volume 3 [html] and read pages 1 – 10 and 93 – 99. Or you could click through it piecemeal:
- The Legacy of Calvin Goodrich [23Jan07]
- Goodrichian Taxon Shift [20Feb07]
- Elimia livescens and Lithasia obovata are Pleurocera semicarinata [11July14]
[2] Goodrich, C. (1934) Studies of the gastropod family
Pleuroceridae - I. Occasional Papers of the Museum of Zoology, University of
Michigan 286:1 - 17.
[3] This is actually a bit frustrating. Looking back on Goodrich’s body of work,
there is almost never anything quotable – some “Aha moment” where the fullness
of his vision is revealed. Like Charles
Darwin.
[4] Dillon, R. T., S. J. Jacquemin & M. Pyron (2013)
Cryptic phenotypic plasticity in populations of the freshwater prosobranch
snail, Pleurocera canaliculata.
Hydrobiologia 709: 117-127. [pdf] For more, see:
- Pleurocera acuta is Pleurocera canaliculata [2June13]
- Pleurocera canaliculata and the process of scientific discovery [18June13]
[6] This is a difficult work to cite. J. B. Burch's North American Freshwater
Snails was published in three different ways.
It was initially commissioned as an identification manual by the US EPA
and published by the agency in 1982. It
was also serially published in the journal Walkerana (1980, 1982, 1988) and
finally as stand-alone volume in 1989 (Malacological Publications, Hamburg,
MI).
[7] Minton, R.L. & C. Lydeard (2003) Phylogeny, taxonomy, genetics, and global
heritage ranks of an imperiled, freshwater snail genus Lithasia
(Pleuroceridae). Molecular Ecology 12:
75 – 87.
[8] The best entry to this complex and long-running theme
would be to read FWGNA Volume 2 [html] in its entirety.
Or for a quick lick at the problem, see my essays:
[10] Dillon, R. T. (2014) Cryptic phenotypic plasticity in
populations of the North American freshwater gastropod, Pleurocera
semicarinata. Zoological Studies 53:31.
[pdf] For more, see:
- Elimia livescens and Lithasia obovata are Pleurocera semicarinata [11July14]
[12] I explored the complex relationship between Pleurocera
simplex and P. gabbiana in East Tennessee in a series of three blog posts in 2016:
- The cryptic Pleurocera of Maryville [13Sept16]
- The fat simplex of Maryville matches type [14Oct16]
- One Goodrich missed: The skinny simplex of Maryville is Pleurocera gabbiana [14Nov16]
- CPP Diary: Yankees at The Gap [4Aug19]









